Researchers Reveal Role of TRIM24 in Antiviral Immunity
A recent study, published online in Journal of Experimental Medicine, revealed the role and mechanism of TRIM24 in antiviral immunity, which could be a novel target for antiviral immunotherapy. It was performed by Dr. XIAO Yichuan's research group from Shanghai Institute of Nutrition and Health (SINH) of the Chinese Academy of Sciences.
RNA virus infection remains a global threat to human health. Upon infection, viral RNA is recognized by host RNA sensors, such as retinoic acid inducible gene-I (RIG-I), then recruited to interact with the downstream adaptor mitochondrial antiviral signaling protein (MAVS), which leads to the activation of associated downstream signal molecules and secretion of IFNα/β and proinflammatory cytokines to suppress virus propagation. However, the molecular events controlling the activation of host antiviral immune signaling remain poorly understood.
Ubiquitination is a type of posttranslational modification that has been proved by numerous studies to play critical roles in antiviral immune. In the study, researchers identified 49 significant upregulated or downregulated genes encoding E3 ligases or DUBs using RNA-sequencing and quantitative PCR and then evaluated the function of those genes in antiviral immunity by interfering those genes’ expression using siRNAs. Finally, researchers found that TRIM24 had an important role in antiviral immunity. TRIM24 upregulated vesicular stomatitis virus (VSV)-induced Ifnb1 expression, dramatically curtailing VSV propagation through in vivo and in vitro assay.
Mechanistic study revealed that VSV infection induced abundant TRIM24 translocation to mitochondria dependent on CRM1, where TRIM24 bound with TRAF3 and directly mediated K63-linked TRAF3 ubiquitination at K429/K436. This modification of TRAF3 enabled its association with MAVS and TBK1, which consequently activated downstream antiviral signaling. Furthermore, virus infection gradually decreased both the mRNA and protein levels of TRIM24 in a time-dependent manner. The reason is that virus-activated IRF3 directly bound to the promoter of TRIM24 and thus suppressed its gene expression, forming a negative feedback loop to restrain overactivation of antiviral signaling. Altogether, this study revealed, for the first time, that TRIM24 acts an important role in antiviral immune, providing potential therapeutic targets for antiviral treatment.
This study entitled "TRIM24 facilitates antiviral immunity through mediating K63-linked TRAF3 ubiquitination" was published online in Journal of Experimental Medicine on April 23, 2020, and was supported by the grants from the National Key R&D Program of China, the Strategic Priority Research Program and the Key Research Program of the Chinese Academy of Sciences, the National Natural Science Foundation of China, CAS Key Laboratory of Tissue Microenvironment and Tumor.
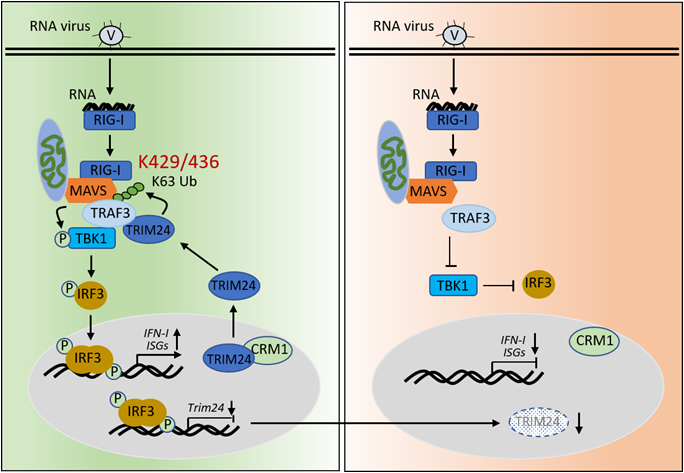
The working model of TRIM24 in mediating antiviral immunity. (Image by Dr. XIAO's group)
Media Contact:
WANG Jin (Ms.)
Shanghai Institute of Nutrition and Health,
Chinese Academy of Sciences
Email: sibssc@sibs.ac.cn
