SMYD2 Drives Mesendodermal Differentiation of Human Embryonic Stem Cells Through Mediating the Transcriptional Activation of Key Mesendodermal Genes
With the ability to differentiate into derivatives of all three germ layers, human embryonic stem cells (hESCs) are developed as an advantageous model for the study of early human embryonic development. Early embryogenesis as well as early hESCs differentiation are finely coordinated by the network of signaling pathways, transcription factors and epigenetic modifications. However, in contrast to the relatively advanced knowledge of signaling pathways and transcription factors, little is known about the contribution of epigenetic modifications, especially, histone methylations, in the regulation of early hESCs lineage differentiation process. SET and MYND domain containing protein 2 (SMYD2) is a histone methyltransferase highly induced during human early mesendodermal differentiation. Knockdown of smyd2a (a homologue gene of human SMYD2) generates severe skeletal and cardiac muscle defects in zebrafish, while cardiac deletion of Smyd2 is dispensable for mouse heart development, suggesting a species-dependent function of SMYD2. However, the precise function of SMYD2 in the regulation of human early lineage fate decision and the underlying mechanism remain to be identified.
Recently, the research group led by Dr.YANGHuangtian, at the CAS Key Laboratory of Tissue Microenvironment and Tumor, Shanghai Institute of Nutrition and Health of Chinese Academy of Sciences found the function of histone methyltransferase SMYD2 in the mesendodermal commitment of hESCs through transcription control of the key mesendodermal signature genes by maintaining H3K4me1 and H3K36me2 at their promoter regions.
Deletion of SMYD2 in hESCs did not affect early neuroectodermal differentiation of hESCs, while it blocked the mesendodermal lineage commitment. This phenotype was rescued by reintroduction of SMYD2 into the SMYD2-/- hESCs. Mechanistically, the bindings of SMYD2 at the promoter regions of critical mesendodermal transcription factor genes brachyury (T), eomesodermin (EOMES), mix paired-like homeobox (MIXL1) and goosecoid homeobox (GSC) were significantly enhanced during mesendodermal differentiation of SMYD2+/+ hESCs but totally suppressed in SMYD2-/- ones. Concomitantly, such suppression was associated with the remarkable reduction of H3K4me1 and H3K36me2 but not H4K20me1 globally and specifically on the promoter regions of mesendodermal genes T, EOMES, MIXL1 and GSC. These results reveal that the histone methyltransferase SMYD2 is dispensable in the undifferentiated hESCs and the early neuroectodermal differentiation, but it promotes the mesendodermal differentiation of hESCs through the epigenetic control of critical genes to mesendodermal lineage commitment.
These findings provide new insights into the function and mechanism of the histone methyltransferase in the hESC early lineage commitment.
The results entitled “SMYD2 Drives Mesendodermal Differentiation of Human Embryonic Stem Cells through Mediating the Transcriptional Activation of Key Mesendodermal Genes” was published online in Stem Cells on July 26th, 2019.
The research was supported by grants from the National Key R&D Program of China, the Strategic Priority Research Program of the Chinese Academy of Sciences, and National Natural Science Foundation of China. We also thank cooperators Prof. Gang Wei from Shanghai Institute of Nutrition and Health.
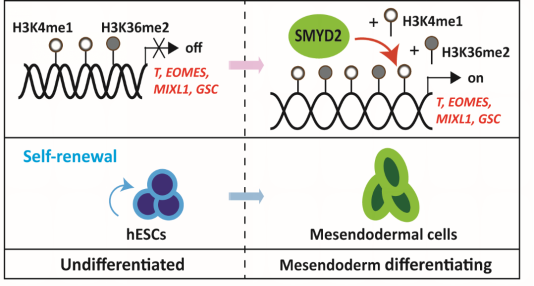
A proposed model for the contribution of SMYD2 to mesendodermal lineage commitment of hESCs. (Image by Dr. YANG Huangtian’s Group)
Media Contact:
WANG Jin (Ms.)
Shanghai Institute of Nutrition and Health,
Chinese Academy of Sciences
Email: sibssc@sibs.ac.cn
