Researchers Reveal Critical Roles of RIPK1 Ubiquitination on Cell Death and Inflammation
RIPK1 is a master regulator of multiple signaling pathways involved in development, inflammation and diverse pathologies. It was initially thought to have a pro-survival role in TNF-induced signaling pathway as Ripk1-deficient mice died soon after birth. Later genetic studies suggested RIPK1 suppressed two cell death pathways, since ablation of Caspase-8/Fadd and Ripk3 allows for normal development and maturation of Ripk1-deficient mice. Remarkably, recent studies have highlighted the critical function of RHIM motif of RIPK1 in preventing ZBP1-dependent necroptosis during development. Formally, a more recent study has highlighted the role of RIPK1 in human disease associated with immunodeficiency, arthritis and intestinal inflammation. These findings indicate that RIPK1 plays critical roles in a variety of biological processes from development to immunity. Although the ubiquitination status of RIPK1 is considered to be critical for regulating RIPK1 functions, the roles for RIPK1 ubiquitination in vivo remains undefined.
A recent study of Dr. ZHANG Haibing’s group from Shanghai Institute of Nutrition and Health of the Chinese Academy of Sciences revealed a critical role of Lys376-mediated ubiquitination of RIPK1 in suppressing RIPK1 kinase activity–dependent lethal pathways during embryogenesis and RIPK3-dependent inflammation postnatally.
In this study, researchers generated mice endogenously expressing RIPK1K376R, a single amino acid change in Lys376 of mouse RIPK1, which is a key docking site for ubiquitin chains. Unexpectedly, mice bearing RIPK1K376R died at embryonic day 12.5 with extensive cell death in fetal liver. However, this lethality was largely prevented by treatment of RIPK1 kinase inhibitor Necrostatin-1, and was fully rescued by concomitant deletion of Fadd and Ripk3 or Mlkl. Importantly, researchers showed that ablation of Tnfr1 rescued the embryonic lethality of Ripk1K376R/K376R mice but caused RIPK3-dependent lethal inflammation in postnatal development. Thus, these findings reveal a crucial role of Lys376-mediated ubiquitination of RIPK1 in suppressing its kinase activity–dependent lethal pathways during embryonic development and RIPK3-dependent inflammation during postnatal development.
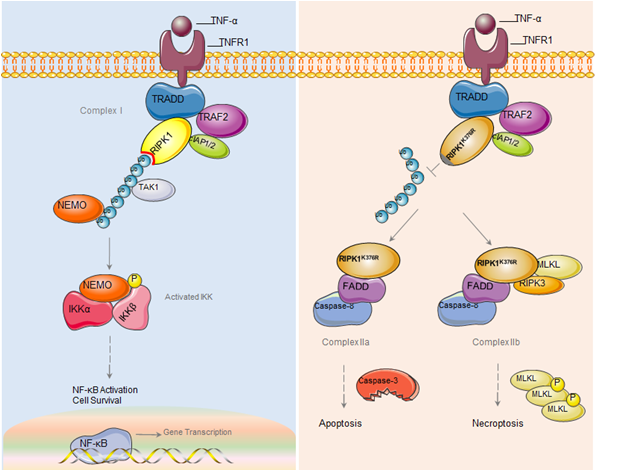
Ubiquitination of RIPK1 suppresses programmed cell death by regulating RIPK1 kinase activation during embryogenesis. (Image by Dr. Zhang's group)
This study unraveled a novel mechanisms by which RIPK1 regulating embryogenesis and postnatal inflammation through Lys376-mediated ubiquitination, which may provide new insights to drug target for clinical prevention and treatment. The research paper entitled "Ubiquitination of RIPK1 suppresses programmed cell death by regulating RIPK1 kinase activation during embryogenesis" was published online on September 13, 2019 in Nature Communications.
Ph.D. student ZHANG Xixi and ZHANG Haiwei are the co-first authors of this work. This work was supported by grants from National Natural Science Foundation of China, Ministry of Science and Technology, and Chinese Academy of Sciences.
Media Contact:
WANG Jin (Ms.)
Shanghai Institute of Nutrition and Health,
Chinese Academy of Sciences
Email: sibssc@sibs.ac.cn
