Scientists Identify a Novel Mechanism of Liver Regeneration via CREB/ATF bZIP Transcription Factor-mediated Inhibition of STAT3
The liver is an important organ of the body that has a central role in maintaining metabolic homeostasis. Although adult hepatocytes are long lived and normally do not undergo cell division, they maintain the ability to proliferate in response to toxic injury and infection. Given the increased number of liver transplant candidates and severe shortage of available organs, an improved understanding of regeneration of the compromised liver would benefit the development of therapeutic approaches to prevent steatosis-related complications and improve the clinical outcome of liver transplantation.
Previous study by Professor LI Yu's research team from Shanghai Institute of Nutrition and Health, Chinese Academy of Sciences revealed CREB/ATF bZIP transcription factor (CREBZF) as a novel regulator in the control of de novo lipogenesis via repressing Insig activity (Zhang F, et al, Hepatology, 2018).
Given that hepatic steatosis is an independent risk factor for liver transplantation, which increases morbidity and mortality, researchers of Prof. Li's group sought to investigate the role of the novel metabolic regulator CREBZF in the progress of liver regeneration.
To uncover the function of CREBZF, scientists characterized CREBZF as a key regulator for liver regeneration. In vivo and in vitro studies demonstrated that CREBZF deficiency stimulates the expression of cyclin gene family and enhances liver regeneration after partial hepatectomy or in response to carbon tetrachloride (CCl4) treatment. Flow cytometry analysis revealed that CREBZF regulates cell cycle progression during liver regeneration in a hepatocyte-autonomous manner. Mechanistically, CREBZF potently associated with the linker domain of STAT3 and repressed its dimerization and transcriptional activity in vivo and in vitro. Importantly, adeno-associated virus mediated overexpression of CREBZF in the liver inhibited the expression of cyclin gene family and attenuated liver regeneration.
The results characterize hepatic CREBZF as a novel coregulator of STAT3 to inhibit regenerative capacity, which may represent an essential cellular signal to terminate regeneration and maintain standard liver mass. These findings may facilitate developing novel strategies to improve liver regeneration and clinical outcome of liver transplantation.
This work was published online in Hepatology on August 30th, 2019, (DOI:10.1002/hep.30919) as a research article entitled “CREBZF as a key Regulator of STAT3 Pathway in the Control of Liver Regeneration in Mice”. This work was supported by grants from Ministry of Science and Technology of China, National Natural Science Foundation of China, Chinese Academy of Sciences, Shanghai Science and Technology Commission, and K. C. Wong Education Foundation.
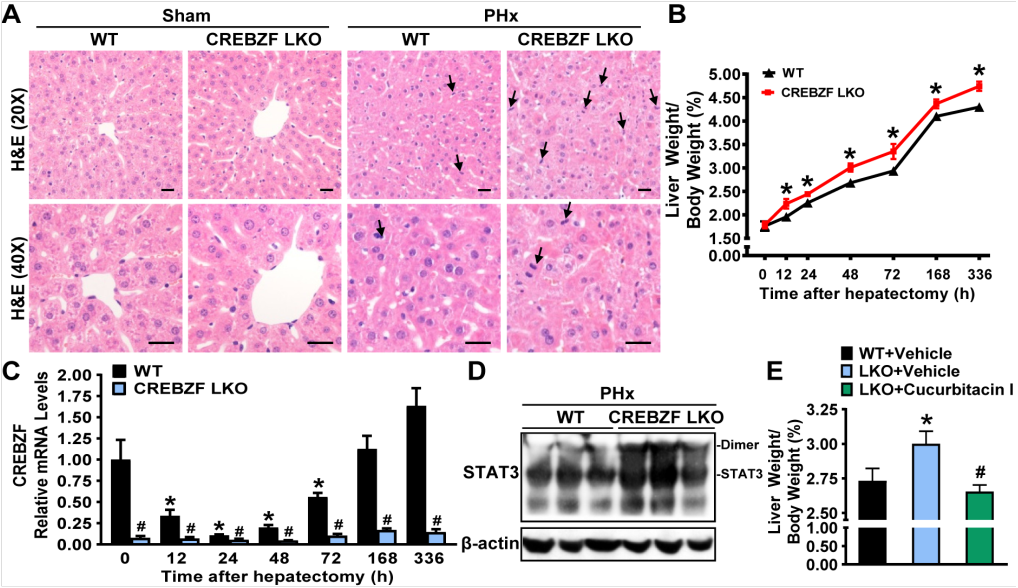
CREBZF regulates liver regeneration via inhibiting STAT3 pathway: potential therapeutic implication in improving liver regeneration and clinical outcome of liver transplantation (Image by Dr. LI Yu’s lab).
Media Contact:
WANG Jin (Ms.)
Shanghai Institute of Nutrition and Health,
Chinese Academy of Sciences
Email: sibssc@sibs.ac.cn
