Hsp90 Inhibition Destabilizes Ezh2 Protein in Alloreactive T cells and Reduces GVHD
Researchers from Dr. ZHANG Yanyun’s lab, Shanghai Institutes for Biological Sciences, CAS revealed for the first time that Ezh2 promotes the alloreactive T cell-mediated GVHD in a H3K27me3-independent mechanism. Inhibiting Hsp90 by its specific inhibitor AUY922 destabilized Ezh2 protein and reduced alloreactive T cell-mediated GVHD, but preserved T cell ability against leukemia, leading to markedly improved survival of mice after allogeneic hematopoietic stem cell transplantation (allo-HSCT).
The recent Blood journal has published the the study entitled “Hsp90 inhibition destabilizes Ezh2 protein in alloreactive T cells and reduces graft-versus-host disease (GVHD) in mice”. This paper has been recommended as a cover art paper.
GVHD remains the major cause of morbidity and mortality after allo-HSCT. GVHD involves complex interactions of immune cells, induction of host-reactive donor effector T cells, and donor T cell-mediated injury to normal tissues. Epigenetic changes have been implicated in T cell-mediated GVHD. Ezh2 is a histone methyltransferase specifically catalyzing the trimethylation of histone 3 at lysine 27. Genetic ablation of Ezh2 in T cells has been shown to reduce alloreactive T cell responses and the subsequent development of GVHD.
To test the translational potential of pharmacological Ezh2 inhibition in GVHD therapy, the authors Dr. Qingrong Huang and et al. found that simply reducing the level of H3K27me3 by administration of the Ezh2 inhibitor GSK126, UNC1999 or EPZ6438, could not prevent GVHD. In contrast, inhibiting Hsp90 by its specific inhibitor AUY922 destabilized Ezh2 protein in T cells, reduced GVHD, and preserved T cell mediated anti-leukemia activity. This effect was accompanied by significantly decreased survival and expansion of alloreactive T cells that produced high levels of IFN-γ and TNF-α in vivo. Introduction of Ezh2 in alloreactive T cells restored their expansion and production of effector cytokines upon AUY922 treatment, suggesting that the impaired T cell alloreactivity by inhibiting Hsp90 is achieved mainly through reducing the protein level of Ezh2.
Thus, these findings identify a previously unrecognized molecular mechanism by which Ezh2 and Hsp90 are integrated to regulate alloreactive T cell responses and GVHD. Targeting the Ezh2-Hsp90 complex using AUY922 represents a novel and clinically relevant approach to reduce GVHD while preserving GVL effects, thereby improving the efficacy of allo-HSCT.
This work was supported by National Natural Science Foundation of China, the Ministry of Science and Technology of China, Strategic Priority Research Program of the Chinese Academy of Sciences, and the Program of Science and Technology Commission of Shanghai Municipality, China.
Current Issue Cover of Blood
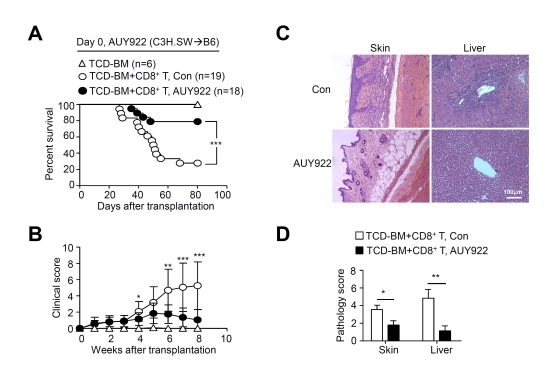
Ezh2-Hsp90 interaction represents an effective target for reducing GVHD:
the Hsp90 inhibitor AUY922 leads to improvement of survival (A), clinical score (B) of GVHD mice and inhibition of inflammation damage to GVHD target organs, such as skin and liver (C, D). (Image provided by Dr. ZHANG Yanyun’s lab)
Author Contact:
ZHANG Yanyun, MD, PhD, Principal Investigator
Shanghai Institutes for Biological Sciences
Chinese Academy of Sciences
Shanghai 200031, P.R. China
Phone: +86-21-54923270
Email: yyzhang@sibs.ac.cn
