- Home >> ALL News >> Highlights
Natural Antiaging Compound Found in Grape Seeds with Lifespan-extending Effects
It was estimated that nearly 1.6 billion individuals worldwide will be older than 65 years of age by 2050. Chronological age is the preeminent risk factor for the leading causes of morbidity and mortality worldwide.
In higher eukaryotes, cellular senescence represents a distinct cell fate in response to a host of extrinsic and intrinsic stimuli. Senescence facilitates embryonic development and wound healing, while restraining carcinogenesis. However, pathologic accumulation of senescent cells in advanced life stage is implicated in organismal aging and age-associated morbidities.
In a study published in Nature Metabolism on Dec. 6th, 2021, researchers from Shanghai Institute of Nutrition and Health (SINH), Chinese Academy of Sciences, conducted a large scale investigation with plant-derived medicinal agents, and revealed that a natural compound in grape seeds, namely procyanidin C1 (PCC1), promotes healthspan and extends lifespan of old mice by interfering with the survival of senescent cells, while leaving normal cells unaffected.
In experimental animals in which senescent cells contribute to age-related physical dysfunction, such as those generated after exposure to ionizing radiation, PCC1 administration significantly decreased the number of senescent cells, reversed organ degeneration and led to health improvements. For mice carrying tumor xenografts, PCC1 also promoted therapeutic outcomes when combined with traditional anticancer agents. Strikingly, PCC1 treatment in a metronomic schedule into very old mice (aged 24-27 months, equivalent to a human age of 75–90 years) increased their remaining lifespan by over 60%, or extended their total lifespan by more than 9%.
Senescent cells produce distinct paracrine and endocrine signals, causing local tissue dysfunction and exerting systemic effects. In this study, Dr. SUN Yu's group at SINH demonstrated that senescent cell removal by apoptosis-inducing "senolytic" agents such as PCC1, effectively limits senescent cell populations in multiple types of tissues and organs, providing benefits in models of geriatric decline and chronic diseases. These data support senescent cells as a pharmacologic target for alleviating effects of fundamental aging processes.
Given the increasing demand for research to develop targeted senotherapeutics addressing the needs of the aging and chronically-ill, the researchers propose that the exact molecular and cellular mechanisms of PCC1 actions warrant continued investigation. "Although PCC1 delivery by intermittent programs appears to be well tolerated by mice in preclinical trials, further efforts are desired to establish safe routes adaptable for PCC1 treatments in humans, key factors that have to be determined by future clinical studies.", Dr. Sun said.
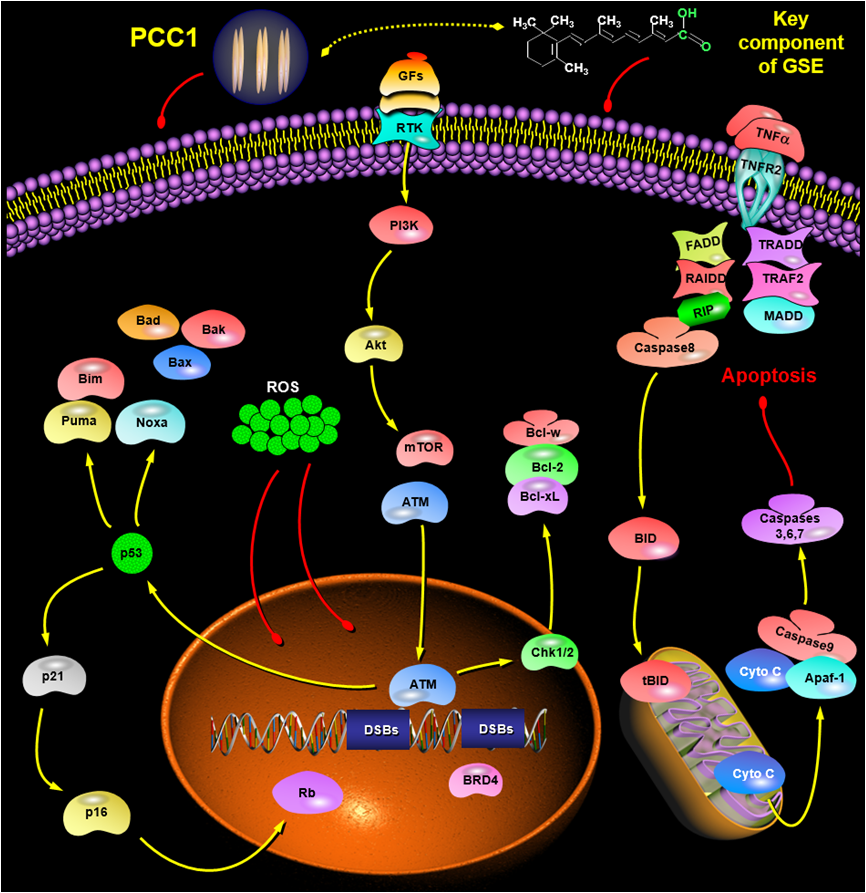
Working model for PCC1 in inducing apoptosis of senescent cells,a process that likely engages multiple signaling pathways. (Image by Dr. SUN Yu's group)
Media Contact:
WANG Jin (Ms.)
Shanghai Institute of Nutrition and Health,
Chinese Academy of Sciences
Email: sibssc@sibs.ac.cn
Web: http://english.sinh.cas.cn/