- Home >> ALL News >> Highlights
Scientists Reveal New DNA-sensing Pathway in CD4+ T Cells and Regulatory Mechanism in Mediating Aging-related Autoimmune Diseases
Aging is a major social issue in China and worldwide and the aging of immune system is a leading cause of chronic inflammation and autoimmune diseases in the elderly individuals. But the specific regulatory mechanism by which aging promotes the development of autoimmune inflammation remains be elucidated. Now, researchers from the Chinese Academy of Sciences (CAS) have revealed that the KU complex mediated-DNA sensing in CD4+ T cells potentiates T cell activation and aging-related autoimmune diseases.
Entitled “Cytoplasmic DNA sensing by KU complex in aged CD4+ T cell potentiates T cell activation and aging-related autoimmune inflammation”, this research was published online in the international academic journal Immunity on March 4, 2021. It is jointly conducted by Dr. XIAO Yichuan's group at the Shanghai Institute of Nutrition and Health (SINH) of CAS and Dr. ZHENG Mingyue's group at the Shanghai Institute of Materia Medica of CAS.
In the elderly individuals, although thymus atrophy causes a decrease in naive T cell output, the number of peripheral T cells does not decrease because of its homeostatic proliferation and activation under the aging state. However, the mechanism by which aging enhances homeostatic proliferation of T cells and thus promotes the development of autoimmune inflammation remains unknown.
In this study, the researchers found that there is a huge accumulation of DNA in the cytoplasm of CD4+ T cells of aged mice and humans, which promotes the proliferation and activation of TCR-induced CD4+ T cells, suggesting that T cells DNA sensing promote their functional activation. The researchers screened the proteins that bind to T cell cytoplasmic DNA by mass spectrometry combined with immunoblotting and found that DNA in T cells does not bind to cGAS, but to KU complex (KU70/KU80).
Further studies revealed that the KU complex was abundantly expressed in the cytoplasm of T cells, and its recognition of DNA in CD4+ T cells promoted the activation of DNA-PKcs. This process in turn mediated the phosphorylation of ZAK at T169. The phosphorylated ZAK then activated the downstream AKT/mTOR pathway, enhancing the proliferation and activation of CD4+ T cells. Thus, activation of the KU complex-mediated DNA-sensing pathway in CD4+ T cells is a key mechanism leading to the development of autoimmune inflammation in aged mice.
The discovery of the newly identified DNA-sensing pathway inspires the researchers to explore potential therapeutic strategies of aging-associated autoimmune inflammation. By using the Caloric Restriction (CR) or Fast-Mimicking Diet (FMD) mouse models, the researchers found that both modes of dieting significantly reduced DNA damage and cytoplasmic DNA accumulation in aged mouse CD4+ T cells, thereby inhibiting ZAK-T169 phosphorylation and activation of downstream AKT/mTOR signaling. The process ultimately suppressed CD4+ T cell activation and aging-associated autoimmune disease.
Furthermore, based on the identified key protein kinase ZAK in the DNA sensing pathway, the researchers applied deep learning combined with molecular simulation to screen a library of approximately 130,000 compounds and obtained iZAK2, a small molecule compound that specifically inhibits ZAK kinase activity. iZAK2 was found to effectively inhibit DNA-induced CD4+ T cell proliferation and activation, thereby alleviating the pathological symptoms of autoimmune disease in aged mice.
Taken together, this study reveals a novel DNA-sensing pathway in aged CD4+ T cells that is independent on cGAS/STING, which promotes T cell activation and proliferation and leads to the development of aging-associated autoimmune diseases. Further investigation and development of inhibitors that block DNA-sensing signaling in T cells may be beneficial for clinical treatment of aging-related autoimmune diseases.
WANG Yan, a PhD student at SINH is the first author of the paper, and Dr. XIAO Yichuan Xiao and Dr. ZHENG Mingyue are co-corresponding authors. The research was supported by projects from the Ministry of Science and Technology, the National Natural Science Foundation of China and CAS.
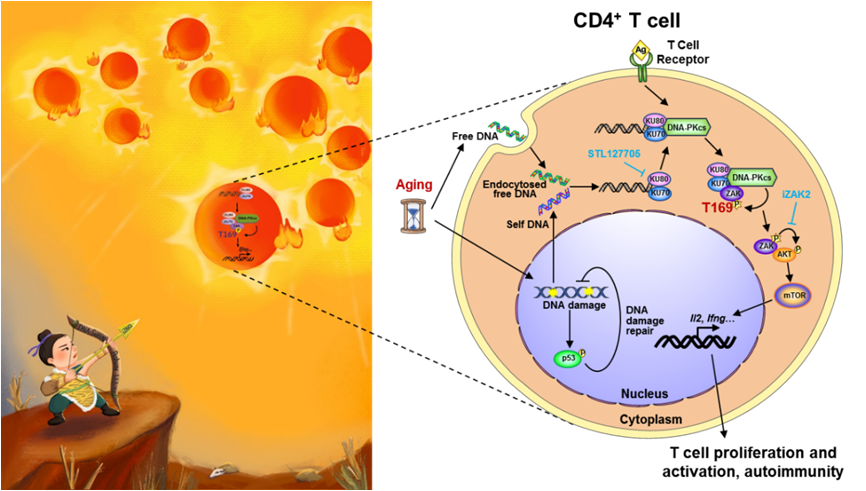
Schematic representation of the cartoon and mechanism of DNA sensing in aged CD4+ T cells promoting its activation and autoimmune inflammation. (Image provided by Dr. XIAO's group)
Media Contact:
WANG Jin (Ms.)
Shanghai Institute of Nutrition and Health,
Chinese Academy of Sciences
Email: sibssc@sibs.ac.cn
Web: http://english.sinh.cas.cn/